Oxidative Stabilization of Olive Oil by Punicalagin During Storage
Rasha A. Mansouri
Abstract
This study aimed to evaluate the antioxidant activity of punicalagin, which is extracted from the pomegranate, on the stability of olive oil in comparison with butylated hydroxytoluene (BHT). The evaluation was carried out by measuring peroxide value (PV) and acid value (AV) to identify the primary products of oil oxidation. Thiobarbituric acid reactive substances (TBARS) and P-Ansidine value (PAV) were used to identify the secondary products of oxidation. Also, the total oxidation index (TOTOX) was calculated to evaluate the overall oxidation status of olive oil samples. Olive oil was divided into three groups: olive oil alone (control), olive oil with BHT, and olive oil with punicalagin. All samples were stored in the same conditions and tested for 60 days. The presence of punicalagin in olive oil significantly reduced peroxides (18.17 Meq/kg oil; p< 0.0001), acid value (5.32 mg KOH/g fat, p< 0.0001), TBARS formation (3.22 ug MDA/g oil, p<0.01), and P-AV (13.82; p<0.0001) in comparison with the control (27.50 Meq/kg oil, 3.67 ug MDA/g oil, and 19.35, respectively). Also, the antioxidant activity exhibited by punicalagin showed similar effects as the synthetic antioxidant, BHT, with no significant differences between them in all parameters. These findings indicate substantial potential for using punicalagin as a natural preservative to improve the quality of oil during storage and to prolong its shelf life.
Key words: natural antioxidant, BHT, oxidation, punicalagin, olive oil
Today, several countries are seeking important compounds and plants that are helpful in the treatment of disease (Ahmad et al., 2018; Sargia et al., 2018), or that can be used in various industries as a natural substance (Esmaielzadeh-Shahrestani et al., 2017). The benefits of using olive oil in one’s daily diet and its role in human health, particularly in preventing cardiovascular disease, inflammation, and cancer, have been extensively reviewed by researchers (Visioli et al., 2006; Cornwell and Ma, 2007). Its high content of fatty acids and other components such as squalene, plant sterols, tocopherols, and polyphenols contribute to the beneficial effects of olive oil. The fatty acid composition of olive oil comprises oleic acid (18:1n–9) 69%, palmitic acid (16:0)14%, linoleic acid (18:2n–6) 12%, and linolenic acid (18:3n–3) <1% (Firestone, 1999).
Despite the favorable effects of fatty acids, unsaturated fatty acids are extremely prone to autoxidation and, therefore, deteriorative changes in the lipid profile of oils can occur, resulting from both in a loss of these benefits and in the development of numerous problems. Oxidized oil is characterized by an off-odor and rancidity, which make the products unacceptable to consumers, resulting in economic losses (Che Man and Tan, 1999). Lipid oxidation is a continuous process; once triggered, it is difficult to terminate, leading to many harmful compounds such as hydroperoxides, which is oxidized further to produce organic compounds. All these compounds are responsible for the deterioration of oil’s nutritional quality and the shortening of its shelf life (Gordon and Kourkimskå,1995).
To enhance the quality of oils and diminish the damaging effects of the peroxidation process, antioxidants are used. Synthetic antioxidants such as butylated hydroxytoluene (BHT) and butylated hydroxyanisole (BHA) are well established for this use (Ali et al., 2013). Nevertheless, consumers’ safety concerns have motivated research to discover a natural source of antioxidants that has the same effect on the oxidation process with a better health profile (Whysner et al., 1994). Therefore, the utilization of natural antioxidants has become a major area of scientific research.
One natural source of antioxidants is the pomegranate due to the presence of tannins, which are high molecular weight phenolic compounds (Omar et al., 2015). The fruit and peel of the pomegranate have been used in herbal remedies in many countries of the world (Reddy et al., 2007; Iqbal et al., 2008). Moreover, pomegranate peel extract has exhibited significant effects as an antimicrobial, antioxidant, and protective agent (Ibrahium, 2010). Pomegranate peel extract has successfully inhibited lipid oxidation in sunflower oil and has been recommended as a natural antioxidant in edible oil to replace commercial antioxidants (Bashir et al., 2016). Pomegranate peel is a potentially valuable by-product as it contains a substantial amount of punicalagin (Aloqbi et al., 2016). An understanding of the antioxidant effect of punicalagin may lead to the utilization of this natural compound to replace synthetic antioxidants. Therefore, the current study aimed to evaluate the effect of punicalagin on the oxidative stability of olive oil during storage and to compare it with the effect of the synthetic antioxidant BHT.
Materials and Methods
Materials
Fresh olive oil was purchased from a local oil press shop. The olive oil was treated either with punicalagin as a natural antioxidant or BHT as a synthetic antioxidant; both types of antioxidants were obtained from Sigma-Alderich (St. Louis, MO).
Oil sample preparation
Oil samples were categorized into three different groups including olive oil alone (control), olive oil with BHT, and olive oil with punicalagin. Concentrated solutions of antioxidants (600 ppm) were prepared by adding 10 mg of BHT and 10 mg of punicalagin separately into 16.6 ml of pyrogallol. The antioxidant was then added to one liter of olive oil. All samples were stored in a dark place at room temperature. An aliquot from each oil sample was withdrawn at 0, 30, and 60 days to monitor the lipid peroxidation process and was prepared in triplicate to obtain independent measurements for each time.
Peroxide values (PV)
Peroxides liberated in the oil samples were determined using an iodometric method established by the Association of Official Agricultural Chemists (AOAC) (1990) with slight modifications. Olive oil samples (5g) were dissolved completely in 30 ml of glacial acetic acid-chloroform solution (2:3 v/v). A saturated potassium iodide solution (0.5 ml) was then added and swirled for one minute followed by the immediate addition of deionized water (30 ml). One ml of starch was added as an indicator. Samples were then titrated against 0.1 N sodium thiosulfate until the blue-gray color disappeared. A blank was also prepared and determined under similar conditions. PV (Meq/kg oil) was calculated as follows:
Peroxide value=
where S = the titrant volume for the oil sample (ml), B = the titrant volume for the blank (ml), N = the normality of the sodium thiosulfate solution, and W = the weight of the oil (g).
Acid values (AV)
The acid value was determined according to Zhang et al. (2010) with some modifications. Briefly, oil samples (1 g) were dissolved in 10 ml of ethanol and shaken very well. The mixture was then titrated against 0.1 N potassium hydroxide (KOH) using phenolphthalein as an indicator until a faint pink color appeared. A blank was also prepared and determined under similar conditions. AV (mg KOH/g fat) was calculated as follows:
Acid value = 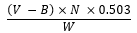

where V = the titrant volume for the oil sample (ml), B = the titrant volume for the blank (ml), N = the normality of KOH, and W = the weight of the oil (g).
Thiobarbituric Acid Reactive Substances (TBARS) values
The concentration of malondialdehyde (MDA) that is produced as a secondary product of lipid hydroperoxide decomposition was measured according to Dasgupta and Klein (2014) with slight modifications. Oil samples (50 ul) were added to test tubes containing a mixture of 0.8 ml of distilled water, 0.2 ml of 8.1% (w/v) sodium dodecyl sulphate, 1.5 ml of 20% (w/v) acetic acid with pH 3.5, and 1.5 ml of 0.8% (w/v) 2-thiobarbituric acid solution in water. The samples were then heated at 100°C for one hour. After cooling, the samples were centrifuged at 4300 x g for 10 min. The absorbance of the upper layer was then measured at 532 nm using a spectrophotometer. The concentration of MDA (μg MDA/g oil) in each sample was then determined by comparing the average optical density of the samples with a standard curve of tetra ethoxy propane (TEP) obtained by using a serial dilution of TEP with a concentration range of 0.2–1.0 μg/ml.
P-Anisidine values (PAV)
PAV values were determined using the standard AOCS (2015) method, which requires the preparation of two solutions. First, solution A was prepared by dissolving oil samples (0.5 g) in 25 ml of 2, 2, 4-trimethylpentane (iso-octane). The absorbance of this solution at 350 nm was measured using iso-octane as a blank. Second, solution B was prepared by dissolving P-anisidine (1 ml) in glacial acetic acid (2.5 g/ L) followed by adding 5 ml of solution A. The solution was mixed and stored against the light for 10 minutes exactly; absorbance was then measured at 350 nm using P-anisidine and iso-octane (1:5 ml) as a blank. The PAV was calculated according to the following equation:
P-Ansidinevalue = 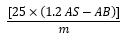
where, 𝐴𝑆 = the 𝑎𝑏𝑠𝑜𝑟𝑏𝑎𝑛𝑐𝑒𝑜𝑓𝑠𝑜𝑙𝑢𝑡𝑖𝑜𝑛𝐵𝑎𝑡 350 𝑛𝑚, 𝐴𝐵 = the 𝑎𝑏𝑠𝑜𝑟𝑏𝑎𝑛𝑐𝑒𝑜𝑓𝑠𝑜𝑙𝑢𝑡𝑖𝑜𝑛𝐴𝑎𝑡 350 𝑛𝑚, and 𝑚 = the weight of the substance to be examined in test solution A (g).
Total oxidation values (TOTOX)
The total oxidation value of the olive oil samples was calculated using Shahidiand Wanasundara’s(2002) method according to the following equation:
𝑇𝑂𝑇𝑂𝑋 = 2 ∗𝑃V + 𝑃𝐴𝑉
where, PV= peroxide value, and PAV= P-Anisidine value.
Statistical analysis
All experimental methods were replicated on at least three separate occasions. The results were statistically analyzed using GraphPad Prism software version 8.1.0. Statistical analysis comparisons were made by two-way analysis of variance (ANOVA) followed by Tukey’s multiple comparison test. Data were presented as means ± SEM, and differences of p ˂ 0.05 were considered to be significant.
Results
Peroxide values
The peroxide values of olive oil alone, olive oil with BHT, and olive oil with punicalagin are shown in Figure 1. At 0 storage time, all samples exhibited low peroxide value with no significant difference between them. During storage, the peroxide value in all samples elevated significantly due to the continuing oxidation process; olive oil alone scored the highest value (27.50 Meq/kg oil) at day 60. From day 30 until the end of the study period, the oil with BHT and the oil with punicalagin showed a significant reduction in peroxide formation when compared with the control. Moreover, the oil with BHT and the oil with punicalagin showed similar peroxide values with no significant difference between them.
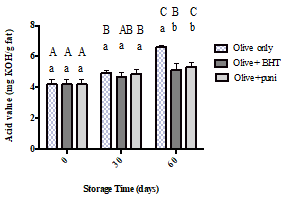
Figure 1:Peroxide values for olive oil alone, olive oil with BHT, and olive oil with punicalagin at 0, 30, and 60 days of storage time. Values correspond to the means ± SEM of three determinations. ANOVA analysis was performed followed by Tukey’s multiple comparisons test. Different uppercase letters indicate a significant difference (p < 0.05) in the peroxide value of the same type of oil at different amounts of storage time. Different lower-case letters mean a significant difference (p < 0.05) between the pure olive oil and the olive oil with antioxidants at the same amounts of storage time.
Acid values (AV)
Acid values increased with increasing amounts of storage time for all the samples, but no regular pattern of elevation could be observed. The control sample exhibited the highest acid value (6.61 mg KOH/ g fat) at day 60. At the same amount of storage time, the oil with BHT and the oil with punicalagin showed significantly lower acid values (5.11 mg KOH/ g fat, p<0.0001 and 5.32 mg KOH/ g fat, p<0.0001, respectively) than the control sample.
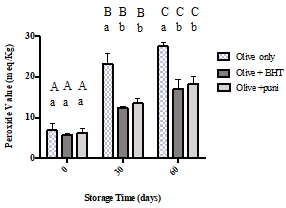
Figure 2:Acid values for olive oil alone, olive oil with BHT, and olive oil with punicalagin at 0, 30, and 60 days of storage time. Values correspond to the means ± SEM of three determinations. ANOVA analysis was performed followed by Tukey’s multiple comparisons test. Different upper-case letters mean a significant difference (p < 0.05) in the acid value of the same type of oil at different amounts of storage time. Different lower-case letters mean a significant difference (p < 0.05) between the pure olive oil and the olive oil with antioxidants at the same amount of storage time.
Thiobarbituric acid reactive substances (TBARS) values
Figure 3 shows the TBARS values for all the examined samples and illustrates that there was a slow increase in TBARS values until day 30 of storage, followed by a slow decrease until the end of the study period. Noticeably, there were significant differences in the results for oil treated with BHT or punicalagin when comparing their values with those of the control sample at day 30 and at the end of the storage time. The difference between values significantly (p<0.0001) appeared from day 30 of storage with a score of 3.04 µg MDA/g oil for the oil with BHT and 3.54 µg MDA/g oil for the oil with punicalagin. The same pattern was observed at day 60 of storage with no significant difference between the BHT and punicalagin samples.
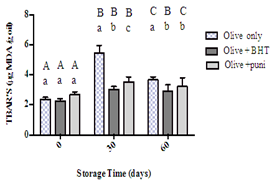
Figure 3:TBARS values for olive oil alone, olive oil with BHT, and olive oil with punicalagin at 0, 30, and 60 days of storage time. Values correspond to the means ± SEM of three determinations. ANOVA analysis was performed followed by Tukey’s multiple comparisons test. Different upper-case letters mean a significant difference (p < 0.05) between the TBARS values of the same type of oil at different amounts of storage time. Different lower-case letters mean a significant difference (p < 0.05) between pure olive oil and olive oil with antioxidants at the same amount of storage time.
P-Anisidine values (PAV)
Figure 4 illustrates the amount of P-anisidine that was measured over the 60 days of the study period. Noticeably, the PAV of the control samples continuously increased until hitting the maximum value at the end of the storage time (19.35). At the beginning of the storage time, there was no significant difference between all samples. A significant difference (p < 0.0001) between the oils with antioxidants and the control started to appear on day 30. At that time, the control exhibited a PAV of 19.0, which is significantly higher compared with the oil with BHT (7.38; p<0.0001) and the oil with punicalagin (12.66; p<0.0001). Similarly, at day 60, the samples with both synthetic and natural antioxidants were able to significantly resist oxidation, scoring 11.50 and 13.82, respectively (both p<0.0001), when compared to the control sample (19.35). However, the oil sample with BHT showed better results compared to the oil sample with punicalagin.
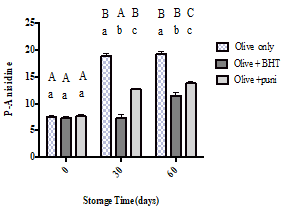
Figure 4:P-Anisidine values for olive oil alone, olive oil with BHT, and olive oil with punicalagin at 0, 30, and 60 days of storage time. Values correspond to the means ± SEM of three determinations. ANOVA analysis was performed followed by Tukey’s multiple comparisons test. Different upper-case letters mean a significant difference (p < 0.05) between the p-ansidine values of the same type of oil at different amounts of storage time. Different lower-case letters mean a significant difference (p < 0.05) between pure olive oil and olive oil with antioxidants at the same amount of storage time.
Total oxidation (TOTOX) values
Figure 5 shows the total oxidation changes over the storage time for the control sample, the oil with BHT, and oil with Punicalagin. As presented in the figure, the oxidation values increased during storage; the control sample hit the maximum value (74.35) at day 60. On the other hand, the oil with BHT and the oil with punicalagin exhibited significantly lower oxidation values at 45.50 and 50.16, respectively, when compared to the control on the same day of storage (p<0.0001).
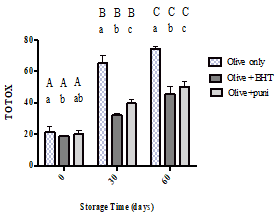
Figure 5: Total oxidation index for olive oil alone, olive oil with BHT, and olive oil with punicalagin at 0, 30, and 60 days of storage time. Values correspond to the means ± SEM of the three determinations. ANOVA analysis was performed followed by Tukey’s multiple comparisons test. Different upper-case letters mean a significant difference (p < 0.05) between the total oxidation index of the same type of oil at different amounts of storage time. Different lower-case letters mean a significant difference (p < 0.05) between the pure olive oil and the olive oil with antioxidants at the same amount of storage time.
Discussion
This study evaluated the effect of punicalagin on the oxidative stabilization of olive oil over a storage period of 60 days. The evaluation was carried out by measuring different parameters including peroxide, acid, TBARS, and p-anisidine values. TOTOX was also calculated to give an overall view of the oxidation status during the storage time.
The effect of punicalagin and BHT on peroxide values
Peroxide value has been used by scientists as an indicator of the primary stage of lipid peroxidation, where lipid radicals are attacked by oxygen to form lipid hydroperoxides (Schwieter, 2009). In this study, a gradual elevation in peroxide values over increasing storage time was observed for all samples. There was a significant difference for the olive oil with punicalagin versus the control after 30 days of storage. Similarly, a significant difference existed for the olive oil with BHT, which scored 12.40 Meq/kg oil, versus the control at the same amount of storage time. Noticeably, there was no significant difference between punicalagin and BHT in their ability to reduce the formation of peroxides, which suggests that punicalagin is as powerful as BHT. Many studies have confirmed that natural antioxidants such as guava leaves (Anwar et al., 2006), ginger (He et al., 2001), and garlic (Iqbal and Bhanger, 2007) have a significant impact on reducing peroxide formation when added to edible oils. The results obtained in this study may be explained by the addition of punicalagin, which exhibited a synergetic effect with the antioxidant compounds that are naturally present in olive oil (Coates et al., 2010; Orsavova et al., 2015). On the other hand, Bashir et al. (2016) illustrated that the addition of punicalagin significantly increased the amounts of primary and secondary oxidation products when added to sunflower oil. It may be hypothesized that the inhibition effect of punicalagin exhibited in the current study may be a result of the phenolic antioxidants inhibiting the lipid oxidation process at the cost of their own life (Anwar et al., 2006).
The effect of punicalagin and BHT on acid values
Oil acidity was used in this study as a parameter to evaluate the quality of olive oil with and without antioxidants by calculating the free fatty acids that were liberated in the system as a result of the hydrolysis of triglycerides (Cornelius, 1966). Generally, the extraction of olive oil followed by a correct crushing procedure results in high-quality oil with low acidity (Gutierrez et al., 2000). However, the degree of hydrolysis might be accelerated by moisture (Iqbal and Bhanger, 2007). The acid values in this study continued to increase with the increase in storage time for all samples, but no regular pattern of the increase could be observed. The control sample exhibited the highest AV at day 60, while the olive oil with BHT exhibited the lowest AV. At the same amount of storage time, the olive oil with punicalagin also exhibited a significant reduction effect on the hydrolysis of triglycerides in comparison with the control. This may be related to the interaction between moisture and punicalagin, revealing less interaction between moisture and olive oil molecules and hence minimization of the hydrolysis process over a longer storage period. The results obtained in the current study agree with those reported by Bashir et al. (2016), who found that pomegranate peel extract stabilized sunflower oil by significantly reducing the percentage of free fatty acid from 0.4667% to 0.2599%.
The effect of punicalagin and BHT on TBARS values
It is well known that a delay in hydroperoxide formation or the production of the secondary products of oxidation can be used to evaluate the efficacy of antioxidants (Chen and Ahn, 1998). Since hydroperoxides are unstable, they rapidly transform into secondary oxidation products (Gray, 1978). PV does not represent the absolute oxidation state of the oil. Therefore, TBARS was conducted in this study to further evaluate punicalagin as a natural antioxidant. As the oxidation process continued, volatile compounds were formed due to the decomposition of lipid peroxide (Frankel, 2011). An accumulation of MDA was observed in this study for all samples with increasing storage time until day 30. These results are the following studies by Iqbal et al., 2008 and Bardhan et al., 2014. The decomposition of MDA and the formation of further oxidation compounds may explain the low value of MDA exhibited by the control and the olive oil with antioxidants at day 60. The addition of either BHT or punicalagin to the olive oil reduced the formation of MDA in these oil samples compared to olive oil alone. Many studies have revealed that the addition of natural antioxidants results in a reduction in MDA formation. For example, pomegranate peel was effective in stabilizing sunflower oil during storage (Bashir et al., 2016). Garlic extract was also used to stabilize sunflower oil and successfully prevent the formation of TBARS (Iqbal and Bhanger, 2007). Similar results were obtained by Goli et al. (2005), in which pistachio hull extract exhibited more oxidative stability on soybean oil than the controls. Martínez et al. (2013) found that using rosemary extract significantly reduced the oxidation of walnut oil under dark storage conditions and improved the oil’s shelf life.
The effect of punicalagin and BHT on P-Anisidine values
Both PAV and TBARS measures the formation of secondary lipid peroxidation, in particular, the formation of carbonyl compounds (Wang et al., 2011). These compounds are responsible for the off-flavor of oxidized oil. In the current study, as the oxidation process continued, carbonyl compounds accumulated, and the control sample hit the maximum P-Anisidine value on day 60 of storage. Punicalagin and BHT significantly lowered the P-Anisidine value in comparison with the control. When comparing the effect of the two types of antioxidants, BHT was found to be more effective than punicalagin. The results obtained in this study are following studies carried out by Li et al., 2012 and Zhang et al., 2018; the latter demonstrated that synthetic antioxidants exhibited a higher activity than natural antioxidants (pecans added to rapeseed oil).
The effect of punicalagin and BHT on the total oxidation index
As the measurement of peroxides alone or carbonyl compounds alone does not represent the absolute oxidation status of studied oil, TOTOX was conducted in this study as it measures both primary (PV) and secondary products (PAV) (Agregán et al., 2017). In this study, a continuous increase in the TOTOX index was observed for all samples as the storage period increased. These results follow a study by Maszewska et al. (2018) as they noticed a significant increase in the oxidation status of edible oils during storage. Starting from day 30 of storage, punicalagin, as well as BHT, significantly lowered the TOTOX index in comparison with olive oil alone. Ibrahium (2010) demonstrated that the presence of pomegranate peel extract (800 ppm) in sunflower oil increased the induction period of oxidation from 8.2 hours (control) to 13.4 hours and exhibited 63.4% antioxidant activity. The results obtained in this study showed the effectiveness of punicalagin in slowing the oxidation process by reducing the formation of both primary and secondary oxidation products. The unique structure of punicalagin with the presence of 15 hydroxyl groups may account for the scavenging activity represented by punicalagin.
Conclusion
The results obtained in this study indicate that punicalagin as a natural antioxidant can exert as a powerful stabilization on olive oil as synthetic BHT. Punicalagin improves the resistance of olive oil against the oxidative rancidity that occurs during storage by significantly inhibiting the formation of peroxides and carbonyl products. Therefore, it can be concluded that punicalagin can stabilize olive oil and prolong its shelf life.
It is recommended that future studies test punicalagin as a natural antioxidant on other oil types. A higher concentration of punicalagin also needs to be examined to evaluate its efficacy in comparison to synthetic antioxidants.
Disclosure statement
No potential conflict of interest was reported by the author.
ORCID
Rasha A. Mansouri https://orcid.org/0000-0003-2698-7475.
References
Agregán, R.; Munekata, P. E.; Domínguez, R.; Carballo, J.; Franco, D.; &Lorenzoa, J.M. (2017). Proximate composition, phenolic content, and in vitro antioxidant activity of aqueous extracts of the seaweeds Ascophyllum nodosum, Bifurcaria bifurcata, and Fucus vesiculosus. Effect of addition of the extracts on the oxidative stability of canola oil under accelerated storage conditions. Food Research International, 99 (3), 986-994.
Ahmad, M. S., Shawky, A., Ghobashy, M. O. &Felifel, R. H. A. (2018). Effect of Some medicinal plants on life cycle of Citrus Brown Mites (Eutetranychusorientalis). International Journal of Pharmaceutical Research & Allied Sciences, 7(4), 13-17.
Ali, H. M.; Abo-Shady, A.; Sharaf Eldeen, H. A.; Soror, H. A.; Shousha, W. G.; Abdel-Barry, O. A.; &Saleh, A. M. (2013). Structural Features, Kinetics, and SAR Study of Radical Scavenging and Antioxidant Activities of Phenolic and Anilinic Compounds. Chemistry Central Journal,7 (1), 53.
Aloqbi, A.; Omar, U.; Yousr, M.; Grace, M.; Lila, MA.; &Howell, N. (2016). Antioxidant Activity of Pomegranate Juice and Punicalagin. Natural Science, 8, 235–46.
Anwar, F.; Jamil, A.; Iqbal, Sh.; &Sheikh, M. (2006).Antioxidant activity of various plant extracts under ambient and accelerated storage of sunflower oil. Grasasyaceites, 57 (2).https://doi.org/10.3989/gya.2006.v57.i2.36
AOAC. (1990). Official methods of analysis (15th ed.). Washington, DC: Association of Official Analytical Chemists.
AOCS Press. (2015). Fats and Oils Handbook. Edited by Bockisch, M; Published by AOCS Press.
Bardhan, J.; Sahoo, B.; Chakraborty, R.; &Raychaudhuri, U. (2014). Effect of addition of rice bran oil extract on the stability of sunflower oil, sesame oil, and their blends. International Food Research Journal, 21(6), 2293–98.
Bashir, Sh.; Rasool, Gh.; Randhawa, M. A.; Sharif, M. K.; Gillani, S. A.; Shah, F. H.; Khan, A. A.;& Ullah, S. (2016). Utilization of Pomegranate Peel Extracts to Enhance the Stability of Sunflower Oil. Pakistan Journal of Food Sciences, 26 (4), 218–225.
Che Man, Y.B.; &Tan, C.P. (1999).Effects of natural and synthetic antioxidants on changes in refined, bleached, and deodorized palm olein during deep‐fat frying of potato chips. Journal of Amer Oil ChemSoc, 76, 331-339. DOI:10.1007/s11746-999-0240-y
Chen, X.; &Ahn, D. (1998).Antioxidant activities of six natural phenolics against lipid oxidation induced by Fe2+ or ultraviolet light. Journal of the American Oil Chemists’ Society,75, Article number: 1717.
Coates, P. M.; Betz, J. M.; Blackman, M. R.; Cragg, G. M.; Levine, M.; Moss, J.;& White, J. D. (2010). Encyclopedia of Dietary Supplements. Second Edition.; published by Informa Healthcare, Paul Street, London, Uk.
Cornelius, J. A. (1966). Some technical aspects influencing the quality of palm kernels. Journal of the Science of Food and Agriculture, 17 (2). https://doi.org/10.1002/jsfa.2740170201
Cornwell, D. G.; &Ma, J. (2007). Studies in vitamin E: biochemistry and molecular biology of tocopherol quinones. Vitam. Horm, 76, 99–134.
Dasgupta, A.; &Klein, K. (2014). Antioxidants in Food, Vitamins, and Supplements. Elsevier Inc.: San Diego, CA, USA.
Esmaielzadeh-Shahrestani, F., Rahnavard, A. &Babakhani, B. (2017). Investigation of Antioxidant and Antibacterial Characteristics in Cydonia Leaves Extract. World Journal of Environmental Biosciences, 6(SI), 1-4.
Firestone, D. (1999).Physical and Chemical Characteristics of Oils, Fats, and Waxes; AOCS Press: Champaign, IL.
Frankel, E. N. (2011). Nutritional and biological properties of extra virgin olive oil.Journal of Agricultural and Food Chemistry. Food Chem, 59 (3), 785-792.
Goli, A. H.; Barzegar, M.; &Sahari, M. A. (2005). Antioxidant activity and total phenolic compounds of pistachio (Pistaciavera) hull extracts. Journal of Food Chemistry, 92 (3), 521-525.
Gordon, M. H.;&Kourkimskå, L. (1995).The effects of antioxidants on changes in oils during heating and deep-frying.Journal of the science of food and agriculture, 68.
GRAY, J.I. (1978). Measurement of lipid oxidation, a review. J. Am. Oil Chem. SOC, 55,539-546.
Gutierrez, F.; Varona, I.; &Albi, M.A. (2000).Relation of acidity and sensory quality with sterol concentration of olive oil from stored fruit. J. Agric. Food Chem, 48,1106–1110. DOI: 10.1021/jf9907337.
He, W.; Li, L.; Li, Y.; Guo, S.;&Guo, B. (2001).Anti-oxidative activity of a new compound from ginger. ZbongguoBingliShenliZashi, 17(5), 461–463.
Ibrahium, M.I. (2010). Efficiency of Pomegranate Peel Extracts as Antimicrobial, Antioxidant, and Protective Agents. World Journal of Agricultural Sciences, 6 (4), 338-344.
Iqbal, S.; &Bhanger, MI. (2007). Stabilization of sunflower oil by garlic extract during accelerated storage. Food Chemistry, 100 (1), 246–54.
Iqbal, S.; Haleem, S.; Akhtar, M.; Zia-ul-Haq, M.; &Akbar, J. (2008). Efficiency of pomegranate peel extracts in stabilization of sunflower oil under accelerated conditions. Food Research International, 41(2), 194–200.
Li, C.; Tang, Z.; Huang, M.; Tao, N.; Feng, B.; &Huang, S. (2012). Antioxidant efficacy of extracts produced from pickled and dried mustard in rapeseed and peanut oils. Journal of Food Science, 77 (4), C394-C400.https://doi.org/10.1111/j.1750-3841.2011.02606.x
Martínez, M. L.; Penci, M. C.; Ixtaina, V.; Ribotta, P.D.; &Maestri, D. (2013). Effect of natural and synthetic antioxidants on the oxidative stability of walnut oil under different storage conditions. LWT - Food Science and Technology, 51 (1), 44–50.
Maszewska, M.; Florowska, A.; Dłużewska, E.; Wroniak, M.; Marciniak-Lukasiak, K.; &Żbikowska, A. (2018).Oxidative stability of selected edible oils.Molecules, 23 (7), 1746.https://doi.org/10.3390/molecules23071746
Omar, U.; Aloqbi, A.; Yousr, M.; &Howell, N. (2015). Protective effects of punicalagin on caco-2 intestine cell line under oxidative stress caused by tert-butyl hydroperoxide. Journal of Pharmacy and Nutrition Sciences,5 (4), 249–56.
Orsavova, J.; Misurcova, L.; Ambrozova, J. V.; Vicha, R.; &Mlcek, J. (2015). Fatty Acids Composition of Vegetable Oils and Its Contribution to Dietary Energy Intake and Dependence of Cardiovascular Mortality on Dietary Intake of Fatty Acids. International Journal of Molecular Sciences, 16 (6), 12871-12890.
Reddy, M., S. Gupta, M. Jacob, S. Khan, & D. Ferreira. (2007). Antioxidant, antimalarial, and antimicrobial activities of tannin-rich fractions, ellagitannins, and phenolic acids from Punicagranatum L. J. PlantaMedica, 73, 461-467.
Sargia, B., Singh, B., Gupta, N., Gahlot, L. K., Gulati, T. &Hasija, Y. (2018). MED-PDB: An online database of medicinal plants. Journal of Advanced Pharmacy Education & Research, 7(4), 204-207.
Schwieter, U. (2009). Á 401 Ñ Fats and Fixed Oils, 1–14.
Shahidi, F.; &Wanasundara, U.N. (2002).Methods for measuring oxidative rancidity in fats and oils. In C. C. Akoh&D. B. Min (Eds.), Food Lipids: Chemistry, nutrition, and biotechnology (pp. 465-487). New York, NY: Marcel Dekker Inc.
Visioli, F.; Bogani, P.; &Galli, C. (2006). Healthful properties of olive oil minor components in olive oil: Chemistry and Technology; Boskou, D., Ed.; AOCS Press: Champaign, IL; pp 173- 190.
Wang, H.; Liu, F.; Yang, L.; Zu, Y.; Wang, H.; Qu, S.;& Zhang, Y. (2011). Oxidative stability of fish oil supplemented with carnosic acid compared with synthetic antioxidants during long-term storage. Food Chemistry, 128 (1), 93–99.
Whysner, J.; Wang, C.X.; Zang, E.; Iatropoulos, M.J.; &Williams, G.M. (1994).Dose-response of promotion by butylated hydroxyanisole in chemically initiated tumors of the rat forestomach. Food and Chemical Toxicology, 32 (30), 215-222.
Zhang, Y.; Yang, L.; Zu, Y. G.; Chen, X. Q.; Wang, F. J.;&Liu, F.(2010). Oxidative stability of sunflower oil by carnosic acid compared with synthetic antioxidants during accelerated storage. Food Chemistry, 118, 656–662.
Zhang, Y.Y.; Zhang, F.; Thakur, K.; Ci, A.T.; Wang, H.; Zhang, J.G.; &Wei, Z.J. (2018). Effect of natural polyphenol on the oxidative stability of pecan oil. Food and Chemical Toxicology, 119, 489-495. https://doi.org/10.1016/j.fct.2017.10.001