Emerging Role of the Fibroblast Growth Factor 23/Klotho Axis in Cardiovascular and Metabolic Conditions: A Narrative Review
Alexander E. Berezin*, Sergii V. Pavlov, Olga P. Pakholchuk
Abstract
Klotho protein is a powerful regulator of various metabolic pathways including endothelial integration, cell adaptation processes, ion transport, signal transduction, regulation of calcium, potassium, parathyroid hormone, phosphates and glucose metabolism, and reduction of oxidative stress. Klotho protein ensures its biological effects through binding with fibroblast growth factor (FGF)-23 and altered Klotho/FGF23 axis was found a core element in the pathogenesis of the cardiovascular and metabolic disease. It has been found that predisposition for CV disease can be related to Klotho gene polymorphism, while the clinical significance of this finding is required to be elucidated in large clinical studies. The aim of the review was to summarize findings regarding the role of the FGF23/Klotho axis in the CV and metabolic conditions, as well as to identify perspectives to use Klotho protein as a promising pharmacological target for personifying therapy.
Key words: Klotho; fibroblast growth factor-23; biomarkers; cardiovascular disease; metabolic disease
Introduction
Previous numerous experimental and clinical studies have established that Klotho protein not only regulates the aging process of the body, but also affects various metabolic pathways in the body, which includes its participation in endothelial integration, cell adaptation processes, ion transport, signal transduction, regulation of calcium, potassium, parathyroid hormone, phosphates and glucose metabolism, and reduction of oxidative stress (Xu and Sun, 2015; Georgiou et al., 2017; Hu et al., 2013; Corsetti et al., 2016). Klotho protein modulates the expression of global transcription factors, such as HSF, GATA, and FoxO. Klotho protein influences the onset of several premature senescent phenotypes in humans and mice, including osteoporosis, stroke, cardiovascular disease, and atherosclerosis. Additionally, the anti-inflammatory, tumor-suppressive properties of Klotho protein have been disclosed (Zhao et al., 2011; Liu et al., 2011). It is known that the gene is located on chromosome 13q12 in humans and the 5th chromosome in mice and is flanked by PDS5B and STARD13 (Xu and Sun, 2017). The human, rat, and mouse Klotho genes contain five exons and four introns that transcribe 3036, 3042, and 3042 nucleotide mRNAs, respectively. Up until now, the genes encode three α-Klotho and 2 β-Klotho forms with known subsequent functions. The aim of the review was to summarize findings regarding the role of the FGF23/Klotho axis in cardiovascular (CV) and metabolic conditions.
Structure of Klotho protein
Klotho protein exists in two forms, including secreted form (α-Klotho protein) and transmembrane (β-Klotho protein). These two forms, in turn, are represented by 3 α-Klotho proteins: a Membrane-bound α-Klotho, a shed α-Klotho, and a secreted α-Klotho (Xu Y, Sun, 2017; Bartali et al., 2013) and 2 forms exist for β-Klotho: a single-pass transmembrane protein and a Klotho-Related Protein – Klrp (Klrp is a transmembrane protein that binds to fibroblast growth factor receptor). A schematic diagram of the Klotho protein biosynthesis is presented in Figure 1.
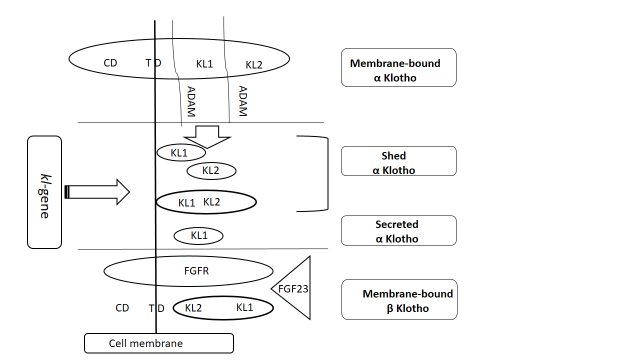
Figure 1. Schematic diagram of α and β-Klotho protein biosynthesis
The synthesis of Klotho protein (α and β form proteins) controls kl-gene. Recent studies have shown the role of hsp-proteins in the protection of kl-gene, Klotho protein and prolongation of its lifespan due to stabilization of the tertiary and quaternary structure (Hui et al., 2017; Olejnik et al., 2018). Membrane-bound α Klotho consists of 3 domains: cytoplasmic (CD), transmembrane (TM), and ectodomain (KL1 and KL2 fragments). With the participation of membrane proteases ADAM 10 and ADAM 17, KL1 and KL2 fragments are cleaved. As a result, shed α-Klotho, and a secreted α-Klotho are formed. α-Klotho protein is secreted into the bloodstream and functions as an endocrine factor (Xu and Sun, 2017). The secretion of α-Klotho protein into the blood, cerebrospinal fluid implements various biological effects: regulation of carbohydrate and phosphate metabolism, inhibition of oxidative stress, antioxidant effects and several others (Xu and Sun, 2015; Shawkat, 2012; Kurosu, 2005; Chang, 2005; Grabner and Faul, 2016). It has been considered α-Klotho protein as a potential biological marker for the prognosis and monitoring of a number of CV and metabolic diseases (Ushigusa et al., 2016; Aiello and Noris, 2010).
The transmembrane β-Klotho protein is represented by the Membrane-bound α Klotho complex with fibroblast growth factor receptor (FGFR) and fibroblast growth factor 23 (FGF23) associated with them by a high-affinity binding. This protein is necessary to increase the affinity of endocrine fibroblast growth (FGF) for FGF receptors (FGFR) in target organs. FGF binds to the receptor, forms a complex, and functions as a paracrine/autocrine factor, while β-Klotho protein regulates energy and mineral metabolism and affects various endocrine factors.
Biological role and function of Klotho protein
The main fundamental biological functions of α-Klotho protein include antioxidant and anti-ageing effects, potential maintenance of normal renal function and angiogenesis, regulates phosphate absorption, calcium and 1,25-dihydroxyvitamin D3 homeostasis, suppression of the insulin signaling pathways, regulation of potassium, phosphate and calcium absorption (Kuro-o, 2013; Kim et a., 2015; Kakareko et al., 2017; Schumann et al. 2016; Zhou et al., 2015).
Antioxidant effects
Interest in the possible health benefits of some compounds has increased in recent years owing to their potent antioxidant and free-radical scavenging activities (Messaoudi et al., 2019; Mourad et al., 2018; Banerjee et al., 2017; Rosidi, 2020). Particular attention should be paid to the antioxidant effects of α-Klotho protein, since due to these properties many other effects are carried out. It was found that Klotho protein is able to regulate the expression of global transcription factors. It has been shown the ability of Klotho protein to increase the FoxO3 phosphorylation (Lim et al., 2017; Yao et al., 2017). FoxO3 is known to regulate the expression of manganese superoxide dismutase (MnSOD), an important enzyme for mitochondrial antioxidant defense in mammalian cells. Overexpression of Klotho protein limits the development of in vitro H2O2-induced apoptosis by reducing the oxidative destruction of DNA, as well as functionally active proteins (enzymes of the antioxidant and thiol disulfide system). It is also due to the modulation of FoxO3 that α-Klotho is able to regulate insulin production.
Glucose and lipid metabolism regulation
Several studies have shown that α-Klotho regulates the signaling pathway of the insulin receptor substrate (IRS) and IGF-1 receptor (IGF-1R) without direct binding to these receptors. It was found that phosphorylated FOXO1, FOXO3a, and FOXO4 remain in the cytoplasm and indirectly increase the rate of glucose production in the liver due to increased transcription of the gene expression encoding the glucose 6-phosphatase enzyme. In addition, by modulating FOXO, α-Klotho can influence adipogenesis by activating CCAAT/enhancer binding proteins (C/EBP). C/EBP belongs to a large family of six conservative transcription factors (α, β, γ, δ, ε, and ζ). Factors C/EBPα, C/EBPβ and C/EBPδ are expressed by brown adipose (BAT) and white adipose (WAT) tissues and regulate adipogenesis phases. C/EBPα acts as an activator for many adipocyte genes, such as type 4 glucose transporter (GLUT4), leptin and adipose tissue-specific fatty acid binding protein (aP2) which is responsible for the level of insulin receptors and the level of one of their primary substrates (IRS-1) (Rosen, 2000). Another possible way of influencing α-Klotho on adipogenesis is its activation of PPARγ2 promoter activity, associated with FOXO, which is necessary for initiation of adipogenesis in the nucleus and restores the activity of preadipocytic factor 1 (Pref-1), one of the epidermal protein-like growth factors, inhibiting adipocyte differentiation. These effects of α-Klotho are accompanied by lipid-lowering effects, increase the expression of adiponectin, which in turn increases tissue affinity to insulin and improves glucose tolerance (Liu et al., 2018; Shen et al., 2018).
Klotho/FGF23 and age-related processes
Klotho's ability to activate PPAR is seen as one of the possible mechanisms for regulation of aging processes. It is known that the expression of PPARα and PPARγ decreases with age. At the same time, activation of PPARα leads to the expression of genes responsible for mitochondrial functions such as capture, activation and β-oxidation of fatty acids, as well as genes encoding the production of antioxidant enzymes - Mn superoxide dismutase and catalase. Non-metabolic effects of PPAR receptors include regulation of tissue inflammation, hypertrophy, oxidative stress, remodeling of extracellular matrix, cell cycle, and angiogenesis (Liu et al., 2018).
The PPAR activation by the Klotho protein associated with the FOXO factor leads to a decrease in oxidative stress, the activity of transcription factors responsible for resistance to oxidative stress increases, and transcription of pro-inflammatory NF-κB is suppressed (Shen et al., 2018). In this regard, the possibility of using recombinant Klotho as a pharmacological agent for enhancing PPARγ activity, leading to an increase in insulin sensitivity, is currently being considered. Klotho protein can be considered as a PPAR agonist (SPPARγMs), capable of influencing the activation or regression of specific groups of target genes in different tissues. In addition, several studies have shown that activation of PPAR by various pharmacological agents leads to parallel activation of the expression and synthesis of Klotho protein, which is apparently associated with their genomic and extragenomic interactions (Zhang et al., 2008).
Another example of the possible effect of α-Klotho on global transcription factors is its inhibitory effect on the expression of the gene encoding nicotinamide adenine dinucleotide phosphatoxidase (NADPH). Also, studies by Wang et al. have demonstrated that Klotho attenuates Angiotensin-II-induced superoxide production, oxidative damage to receptors, lipid, protein molecules of cell membranes, and possible penetration through cAMP/PKA (Wang et al., 2012). It has been shown that Klotho deficiency in mice leads to intensification of oxidative stress and the ROS generation. On the other hand, in vivo activation of the Klotho gene reduces NADPH- and Angiotensin-II-mediated production of superoxide radicals, a decrease in the oxidative stress phenomena, and the progression of hypertension (Izbeki et al., 2010).
Klotho protein and apoptosis
The anti-apoptotic function of Klotho protein is directly related to the antioxidant activity. Klotho protein increases resistance to oxidative stress and apoptosis in COS, HeLa, CHO cells, and HUVECs (Sosa-Pineda et al., 1997). A recent study showed that Klotho protein reduced apoptosis, and the levels of Caspase-3 and Caspase-9 activity and of p53 and p21 expression in HUVECs (Sosa-Pineda et al., 1997). Another study concluded that Klotho regulates the cell lifespan through p53 and p21 proteins, and the relation between Klotho and p53 may be associated with increased signaling through the insulin/IGF-1 pathway in MRC-5 human primary fibroblast cells (Izbeki et al., 2010; Sosa-Pineda et al., 1997). Another study concluded that Klotho regulates the cell lifespan through p53 and p21 proteins, and the relation between Klotho and p53 may be associated with increased signaling through the insulin/IGF-1 pathway in MRC-5 human primary fibroblast cells (Xu and Sun, 2015).
Previous studies have shown that knocking out the Klotho gene led to increased apoptosis in ad-kl-vector-transfected mIMCD3 cells, as well as inhibition of the HSP70 proteins expression and synthesis. The gene encoding HSP70 is probably the major gene related to the anti-apoptotic effect of Klotho. HSPs have a cytoprotective function as molecular chaperones, and their expression increases in response to a wide variety of stresses, including heat shock, hypoxia, exposure to hydrogen peroxide and ischaemia. HSP70 proteins are able to protect cells from toxic and stress factors, and stabilize the actin cytoskeleton under conditions of oxidative stress. Apparently, most of these functions of HSP70 are associated with chaperone activity, i.e., with its ability to recognize damaged or newly synthesized polypeptides and to straighten their structure in an ATP-mediated manner or to remove defective proteins through the proteasomal apparatus. The presented synergism of the HSP 70 and Klotho proteins functioning is primarily due to their general biological properties of chaperone proteins and the anti-age functions. On the one hand, due to its chaperone function, HSP70 proteins under conditions of oxidative stress stabilize the Klotho molecule; on the other hand, Klotho stimulates the entry of HSP proteins into the cell during the intensification of free radical oxidation processes, protecting the structural and functional integrity of macromolecules under the toxic effects of free radicals; also due to this mechanism its anti-age function is implemented (Sugiura et al., 2010; Sugiura et al., 2005; Pavlov et al., 2014). Finally, it is important to note that the antioxidant activity of Klotho plays a significant role in its general biological function of anti-aging, since the role of free radicals in the aging mechanisms of the body is known.
Impaired signaling pathways in FGF23/Klotho system
The analysis of numerous literary sources indicates the pleiotropic nature of the Klotho protein effects, which is directly related to the molecular biology of this protein. As described above, the three forms of Klotho protein have different functions. The membrane Klotho protein forms a complex with the fibroblast growth factor receptor, which is a coreceptor for FGF23. Secreted Klotho protein is involved in the regulation of oxidative stress, being a growth factor and serving to maintain ionic homeostasis. The intracellular forms of Klotho protein inhibit inflammation and aging. Secreted forms of Klotho protein are involved in the regulation of nitric oxide (NO) synthesis and endothelial permeability (Doi et al., 2011). Overexpression of the secreted Klotho protein prolongs the life of animals by inhibiting the reactive oxygen species generation. In addition, Klotho protein binds Wnt signaling proteins and inhibits the activity of their endogenous and exogenous forms. Klotho protein inhibits the development of metastases, as well as the action of some growth factors, including TGF-β1 (Doi et al., 2011; Kuznik et al., 2017; Kim et al., 2015; Kim et al., 2015). It was also found that co-receptors of α-Klotho or β-Klotho are required for endocrine FGFs to act on FGFRs as cofactors (Fujita et al., 2016).
Endocrine fibroblast growth factor 23 (FGF23) is a circulating phosphate-containing hormone whose gene is located on the 12th chromosome. It consists of 3 exons. FGF23 includes amino-terminal sequences of the signal peptide (residues 1–24), central sequences (residues 25–180), and carboxyl-terminal sequences (residues 181–251). The half-life of FGF23 in the blood of healthy people is 58 minutes. In order to exhibit specific activity, FGF23 is bound in the kidney to the FGF receptor by Klotho complexes, after which it stimulates urinary excretion of phosphorus, inhibiting the synthesis of 1,25-dihydroxyvitamin D (So et al., 2013).
Under the influence of FGF-23, the level of calcitriol decreases, which contributes to an increase in the secretion of parathyroid hormone (PTH). If the concentration of FGF23 decreases, the level of calcitriol and Klotho protein increases. The secretion of FGF23 is locally regulated in the bones with the participation of dentin-1 and phosphate-regulating endopeptidase. The complete absence of FGF23 protein disrupts bone mineralization, despite an adequate level of phosphorus and vitamin D in the blood (Kuznik et al., 2017; Fujita et al., 2016; So et al., 2013).
FGF23 is also associated with dyslipidemia, such as increased levels of triglycerides, high density lipoprotein cholesterol deficiency, hyperinsulinemia, HOMA index (homeostatic model assessment), high body mass index, and abdominal obesity (Fujita et al., 2016). With a moderate decrease in renal function, phosphorus excretion is suppressed, which leads to an increase in its content in the blood and an increase in the FGF23 synthesis (So et al., 2013; Wesseling-Perry, 2010). It has been found that FGF23 plays a significant role in preventing premature aging by maintaining the balance of vitamin D. This process is carried out by the interaction of FGF23, vitamin D, the Klotho receptor, and the Klotho gene (Kuznik et al., 2017; So et al., 2013; Wesseling-Perry, 2010).
Accelerating premature aging and altered FGF23/Klotho axis
It has now been established that Klotho protein is one of the most important suppressive genes. Meanwhile, the αKlotho concentration in serum significantly decreases with age. In addition, an inverse correlation ratio was found between age and FGF23, which indicates the role of this factor in the prevention of premature aging. There are negative correlations between the content of FGF23, age, and body mass index, both in healthy people and patients with type 2 diabetes mellitus (Wesseling-Perry, 2010).
Knocking out the Klotho gene is known to cause systemic disorders that accelerate the aging process, as well as cardiovascular diseases. On the contrary, activation of the expression activity of the Klotho gene leads to a decrease in angiolithic degeneration, reduces the possibility of developing atherosclerosis, and prevents the development of endothelial dysfunction (Dalton et al., 2017). Moreover, high concentrations of Klotho protein factor in plasma are associated with a lower likelihood of cardiovascular disease in the elderly (Dalton et al., 2017; Jüppner, 2011).
Vitamin D homeostasis, FGF23/Klotho protein and CV disease
The role of FGF23 in regulating the metabolism of phosphate and vitamin D is widely known; meanwhile, Klotho protein, increasing the NO synthesis, softens the effect of phosphate and FGF23 on the contractility of smooth muscle cells in blood vessels and protects them from the effects of their toxic concentrations (Kuznik et al., 2017; Jüppner, 2011). There is evidence regarding the pivotal role of Klotho and FGF23, showed that they are essential companions of pathological processes such as vascular endothelial dysfunction, diffuse vascular calcification and cardiac hypertrophy, diabetes mellitus, and arterial hypertension. These processes proceed against the background of a decrease in the expression activity of the Klotho gene with parallel overexpression of the p16 and p53 genes. The presented molecular shifts initiate an increase in the level of endothelin-1 and enhance the processes of inflammation, which exacerbates the course of endothelial dysfunction. In addition, the Klotho/FGF23 axis is involved in angiolithic degeneration due to deficiency of active vitamin D, hypercalcemia and hyperphosphatemia. Damage to the heart with a decrease in the expression of Klotho factor can be mediated by a violation of the TRPC6 receptor activity and changes in the electrolyte composition (Ding and Ma, 2015). Moreover, a number of studies have shown the relationship of Klotho gene polymorphism with various cardiovascular diseases (Kim and Jeong, 2016; Zhu et al., 2019; Yokoyama et al., 2019; Donate-Correa et al., 2016).
At present, a variant of the Klotho gene - Klotho Valin-Serin (KL-VS) - a variant of the haplotype, which consists of two amino acids F352V and C370S and can be identified as a single SNP, has been identified. The presented allelic gene is common in the European population and contains three mutations. One of the mutations is “dornant,” the others are associated with the F352V and C370S mutations, which modulate the Klotho protein metabolism. The presence of such SNPs as G-395a, rs564481 (C1818T) and rs9536314, according to experimental and clinical studies, determines lifespan, cardiovascular system, osteoporosis. According to some literature sources, 20% of the European populations are the KL-VS genetic variant carriers, which increase the level of circulating Klotho protein, which affects the life span and determines its tissue protective effects (Kim and Jeong, 2016; Zhu et al., 2019). In this regard, a number of studies are considering the possibility of using the circulating form of Klotho protein as a biological marker of aging, a marker for predicting cardiovascular diseases, and also a new pharmacological agent (Xu and Sun, 2015; Corsetti et al., 2016; Olejnik et al., 2018).
CV disease predisposition and Klotho protein gene polymorphism
Genetic studies have shown the relationship of Klotho gene polymorphism with lifespan, the occurrence of cardiovascular and cerebral catastrophes, and bone destruction. These processes are directly related to changes in the regulation of intracellular biochemical pathways, which includes monitoring the Silent Information Regulator 2 proteins activity, lipid profile, glucose metabolism. According to modern concepts, the basis of the cellular aging mechanism is a violation of the activity of Silent Information Regulator 2 proteins, as a result of which toxic derivatives of free radical oxidation accumulate in the cell and inflammatory processes develop. It was found that the presence of one copy of the Klotho genetic variant (KL-VS) determines the increase in lifespan and limits the development of cardiovascular and cerebral catastrophes. At the same time, two copies of the Klotho gene (KL-VS), on the contrary, determine a decrease in lifespan, increase the risk of developing myocardial infarction and cerebral ischemia. However, lack of the KL-VS variant in the Korean population and the presence of two others, G-395A and C1818T, which were associated with CV diseases and osteoporosis (Zhu et al., 2019).
Klotho protein promotes insulin resistance in pre-diabetics and diabetics
The participation of Klotho protein in anti-aging mechanisms explains its effect on glucose metabolism, insulin synthesis, as well as its regulation of insulin-resistant conditions. According to modern concepts, an insulinlike signaling pathway occupies a special place in genetics. The binding of insulinlike growth factor (IGF-1) to the insulin receptor/IGF-1 on the outer surface of the cell activates phosphoinositol-3-kinase (PI3K) on the inner membrane of the cell, which leads to the formation of a low molecular weight mediator - phosphoinositide-3,4,5-triphosphate. It binds to another kinase called 3-phosphoinositide-dependent kinase 1 (PDK-1) and, in turn, activates (phosphorylates) Akt/PKB and SGK-1 kinases, which allows normal cell growth processes to occur. At the same time, stress tolerance factors, such as the transcription factor FOXO, are turned off. Long-living people have increased insulin sensitivity while maintaining its low level in plasma (Bär et al., 2018; Cheng et al., 2005).
The activity of insulin-like signaling and the expression level of insulinlike peptides are reduced in long-living nematode worms, mice and humans. Heterozygous mice and humans with mutations in the IGF‑1 receptor gene have a longer lifespan. Mutations in the genes of substrates 1 and 2 of the insulin receptor lead to an increase in the lifespan of fruit flies and mice. Mutations in the PI3K, AKT/PKB, PDK kinase genes are accompanied by an increase in the lifespan of animals (Xu Y, Sun, 2015; Chihara et al., 2006; Armoni et al., 2006). In contrast, the activity of PTEN, SHIP1, and SHIP2 phosphatases, which oppose PI3K, promotes longevity. Insulinlike signaling suppresses the activity of stress response mechanisms associated with the FOXO transcription factor. The activity of FOXO and FOXO-dependent genes (PEPCK, Hsps, MnSod) leads to an increase in lifespan. Another FOXO-dependent gene, GADD45, with overexpression also leads to increased lifespan and stress resistance in fruit flies, and is also associated with many age-dependent pathologies in humans. A mutation in the Klotho kidney hormone gene leads to a decrease, and overexpression leads to an increase in the lifespan of mice. Klotho inhibits the effects of the insulin/IGF‑1 pathway, increasing resistance to oxidative stress at the cell and body levels, thereby contributing to longevity. A number of studies have reported that impaired α-Klotho expression reduces insulin production and increases insulin sensitivity. Α-Klotho knockout mice also show less energy storage and lower energy expenditures compared to wild-type mice (Lorenzi et al., 2010; Julia et al., 2017).
The ability of Klotho protein to regulate insulinlike signaling, according to some authors, is associated with distribution proteins. Activated IRS leads to activation of the downstream PI3K/Akt signaling pathway and FOXO phosphorylation. FOXO1 indirectly increases the rate of glucose production in the liver by increasing transcription of glucose-6-phosphatase expression. It has been suggested that FOXO1 negatively regulates adipogenesis by its binding to the PPARγ promoter region, which is necessary for initiating adipogenesis in the nucleus. Other studies have shown that α-Klotho can promote adipogenesis through CCAAT/enhancer binding proteins (C/EBPs) and PPARγ, which together support the expression of adipocyte genes to stop the preadipocytes differentiation. In addition, PPARs are ligand-inducible transcription factors that belong to the superfamily of nuclear hormone receptors. By binding to the retinoid X receptor as a partner, they initiate the transcription of genes containing a specific DNA sequence called Peroxisome Proliferator Response Element (PPRE). The PPAR ligands are fatty acids or their derivatives. PPAR is expressed in tissues where a high level of mitochondrial fatty acid oxidation is required, that is, in the liver, kidneys, heart, skeletal muscle and vascular wall. It is activated by fatty acids, eicosanoids, 15‑d-prostaglandin, and oxidized fatty acids and regulates the expression of genes that promote lipid oxidation and lipoprotein metabolism (for example, the main high-density apolipoprotein Apo A‑1). Thus, PPAR counteracts metabolic syndrome and aging in general (Moskaljev, 2013; Lin and Sun, 2015).
Experimental and clinical studies have reported a decrease in the expression and synthesis of the Klotho gene and protein in patients with type 2 diabetes mellitus (T2DM) and in db/db mice, a T2DM animal model. In addition, it was shown that overexpression of the Klotho gene, on the contrary, weakens the development of diabetes in db/db mice (Lin and Sun, 2015; Hum et al., 2019). Increased β-cell expression of Klotho reduced hyperglycemia and insulin resistance. The Klotho β-cell over-expression contributed to a decrease in the level of intracellular superoxide, and also limited the processes of apoptotic death of β cells in the pancreatic islets. These processes occurred against the background of increased expression of the insulin transcription factor Pdx-1, PCNA marker of cell proliferation and LC3 autophagy in pancreatic islets in db/db mice. These results indicate that β-cell specific expression of Klotho improves β-cell function and weakens the development of T2DM. Therefore, Klotho in vivo expression may offer a new β-cell protection strategy in T2DM (Hum et al., 2019).
FGF23 Klotho system and low-grade inflammation
Except classical functions of the Klotho protein related to premature aging, including multiple organ dysfunction and a significantly shortened life span, recent studies showed it possible participation in the immune reactions. It was found that FGF23 Klotho system has role in the systemic inflammation and impaired function of the immune system in different chronic diseases. Pro-inflammatory cytokines, such as tumor necrosis factor alpha (TNF-α), interleukin (IL)-6 and IL-1β, are potent inducers of FGF23 production. As a consequence of these factors promoting the uncontrolled production and excess levels of FGF23 in circulation elevate the production of various cytokines in diverse tissues, liver, which in turn, amplifies systemic inflammation. Loss of Klotho expression causes increased airway inflammation in mice (Krick et al., 2018). FGF23 can directly stimulate hepatic secretion of inflammatory cytokines (C-reactive protein) via activating FGF receptor isoform 4 (FGFR4), and stimulated calcineurin signaling. Administration of an isoform-specific FGFR4 blocking antibody reduced hepatic and circulating levels of C-reactive protein in the 5/6 nephrectomy rat model of chronic kidney disease (Singh et al., 2016). Whether FGF23 and Klotho-mediated FGFR activation are core elements in pathophysiologic mechanisms in low-grade inflammation remains unclear (Berezin and Berezin, 2019; Krick et al., 2017; Courtney et al., 2004). Alongside these novel relationships between FGF23 and consequential interleukins, Han et al. further expanded the role of FGF23 in inflammation by revealing FGF23 targets peritoneal macrophages to upregulate TNF-α production (Han et al., 2016; Czaya and Faul, 2019).
Despite promising results of the primary studies of the Klotho regulating mechanisms of systemi and local inflammation, still there are no evidences of the ability of the immune cells to express Klotho as it is mainly expressed in the kidney, brain, and parathyroid gland. Whether the macrophages and neutrophils, as well as epithelial cells in vessels and lungs, express Klotho, or not, is currently under debate. It still may have pleiotropic effect as the ectodomain of Klotho can also exist in a soluble form (soluble Klotho, sKL) (Richter and Faul, 2018). Several in vitro studies in multiple different cell types, such as fibroblasts, endothelial cells, vascular smooth muscle cells, cardiac myocytes, pulmonary epithelial cells, oligodendrocytes, and neurons, indicate that sKL has cell-protective activities, including the inhibition of apoptosis, oxidative stress, senescence, and pathologic gene programs, suggesting that sKL might protect against cellular dysfunction as well as myocardial fibrosis and inflammation (Richter and Faul, 2018).
However, it is unclear how one particular protein can inhibit various signal mediators and receptors that significantly differ in their structure, biophysical features, and mode of action. One hypothesis was proposed basing on the in vivo findings showing that raft-associated, but not raft-independent, phosphoinositide 3-kinase (PI3K) signaling is elevated in mice lacking Klotho. Binding sialic acids in glycolipids and glycoproteins is a plausible mechanistic explanation for sKL’s pleiotropic actions (Dalton et al., 2017) sKL preferentially interacts with lipid raft domains where gangliosides, such as GM1 and GM3, in cell membranes are enriched. The association with sKL might then affect overall lipid raft dynamics and composition, thereby regulating the localization and activity of a variety of raft-associated proteins, including signaling receptors and ion channels (Dalton et al., 2017).
Other mechanism is related to the activation of the immune cells by enhanced reactive oxygen species generation. In turn, monocytes and macrophages can release reactive oxygen species at the site of inflammation, which promotes the genome-/epigenome activation of pro-inflammatory genes and shift to the persistency.
In conclusion, the role of Klotho in protecting the structural integrity of many receptors, in particular insulin, reproductive, choline, adrenergic receptors, has clearly been defined. Klotho's antioxidant mechanisms are to some extent associated with its inhibition of FoxO3 phosphorylation, as well as genes encoding the synthesis of antioxidant defense enzymes including superoxide dismutase, catalase, and glutathione peroxidase. Synergy of the fundamental functions of both Klotho protein and HSP-proteins determine the relevance of studies of the conjugate biological effects of these proteins and the establishment of their role in the pathogenesis of CV and metabolic diseases, molecular diagnostics, as well as the development of new potential targets for point-of-care. Probably, Klotho protein could be a promising pharmacological target for personifying therapy in the future. Standardization of analytic technologies is required for detection of the biomarker by the enzyme immunoassay in various biological fluids.
Acknowledgments
Funding and grants: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interest:
Authors have no conflict of interest.
Funding and grants:
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
conflict of interests:
Authors have no conflict of interests.
References
Aiello, S., & Noris, M. (2010). Klotho in acute kidney injury: biomarker, therapy, or a bit of both?. Kidney international, 78(12), 1208-1210.
Armoni, M., Harel, C., Karni, S., Chen, H., Bar-Yoseph, F., Ver, M. R., ... & Karnieli, E. (2006). FOXO1 represses peroxisome proliferator-activated receptor-γ1 and-γ2 gene promoters in primary adipocytes A NOVEL PARADIGM TO INCREASE INSULIN SENSITIVITY. Journal of Biological Chemistry, 281(29), 19881-19891.
Banerjee, S., Mallick, M. A., & Pathade, G. R. (2017). Comparison of Antioxidant activity of in vivo and in vitro leaf explants of Piper longum. J Adv Pharm Edu Res, 7(3), 323-325.
Bär, L., Feger, M., Fajol, A., Klotz, L. O., Zeng, S., Lang, F., ... & Föller, M. (2018). Insulin suppresses the production of fibroblast growth factor 23 (FGF23). Proceedings of the National Academy of Sciences, 115(22), 5804-5809.
Bartali, B., Semba, R. D., & Araujo, A. B. (2013). Klotho, FGF21 and FGF23: novel pathways to musculoskeletal health?. The Journal of frailty & aging, 2(4), 179.
Berezin, A. E., & Berezin, A. A. (2019). Impaired function of fibroblast growth factor 23/Klotho protein axis in prediabetes and diabetes mellitus: Promising predictor of cardiovascular risk. Diabetes & Metabolic Syndrome: Clinical Research & Reviews. 13(4):2549-2556. doi: 10.1016/j.dsx.2019.07.018.
Chang, Q., Hoefs, S., Van Der Kemp, A. W., Topala, C. N., Bindels, R. J., & Hoenderop, J. G. (2005). The ß-glucuronidase klotho hydrolyzes and activates the TRPV5 channel. Science, 310(5747), 490-493.
Cheng, C. L., Gao, T. Q., Wang, Z., & Li, D. D. (2005). Role of insulin/insulin-like growth factor 1 signaling pathway in longevity. World journal of gastroenterology: WJG, 11(13), 1891.
Chihara, Y., Rakugi, H., Ishikawa, K., Ikushima, M., Maekawa, Y., Ohta, J., ... & Ogihara, T. (2006). Klotho protein promotes adipocyte differentiation. Endocrinology, 147(8), 3835-3842.
Corsetti, G., Pasini, E., Scarabelli, T. M., Romano, C., Agrawal, P. R., Chen-Scarabelli, C., ... & Flati, V. (2016). Decreased expression of Klotho in cardiac atria biopsy samples from patients at higher risk of atherosclerotic cardiovascular disease. Journal of geriatric cardiology: JGC, 13(8), 701.
Courtney, J. M., Ennis, M., & Elborn, J. S. (2004). Cytokines and inflammatory mediators in cystic fibrosis. Journal of Cystic Fibrosis, 3(4), 223-231.
Czaya, B., & Faul, C. (2019). The Role of Fibroblast Growth Factor 23 in Inflammation and Anemia. International journal of molecular sciences, 20(17), 4195. doi: 10.3390/ijms20174195
Dalton, G. D., Xie, J., An, S. W., & Huang, C. L. (2017). New insights into the mechanism of action of soluble klotho. Frontiers in endocrinology, 8, 323. doi: 10.3389/fendo.2017.00323
Dalton, G., An, S. W., Al-Juboori, S. I., Nischan, N., Yoon, J., Dobrinskikh, E., ... & Birnbaumer, L. (2017). Soluble klotho binds monosialoganglioside to regulate membrane microdomains and growth factor signaling. Proceedings of the National Academy of Sciences, 114(4), 752-757. doi:10.1073/pnas.1620301114.
Ding, H. Y., & Ma, H. X. (2015). Significant roles of anti-aging protein klotho and fibroblast growth factor23 in cardiovascular disease. Journal of geriatric cardiology: JGC, 12(4), 439.
Doi, S., Zou, Y., Togao, O., Pastor, J. V., John, G. B., Wang, L., ... & Takahashi, M. (2011). Klotho inhibits transforming growth factor-β1 (TGF-β1) signaling and suppresses renal fibrosis and cancer metastasis in mice. Journal of Biological Chemistry, 286(10), 8655-8665.
Donate‐Correa, J., Martín‐Núñez, E., Martínez‐Sanz, R., Muros‐de‐Fuentes, M., Mora‐Fernández, C., Pérez‐Delgado, N., & Navarro‐González, J. F. (2016). Influence of Klotho gene polymorphisms on vascular gene expression and its relationship to cardiovascular disease. Journal of cellular and molecular medicine, 20(1), 128-133.
Fujita, Y., Makishima, M., & Bhawal, U. K. (2016). Differentiated embryo chondrocyte 1 (DEC1) is a novel negative regulator of hepatic fibroblast growth factor 21 (FGF21) in aging mice. Biochemical and biophysical research communications, 469(3), 477-482.
Georgiou A., Lisacek-Kiosoglous A., Yiallouris A., Stephanou A., Patrikios I. Klotho, (2017). The Protein of Faith. EC Neurology. 7 (5), 189-223.
Grabner, A., & Faul, C. (2016). The Role of FGF23 and Klotho in Uremic Cardiomyopathy. Current opinion in nephrology and hypertension, 25(4), 314. doi: 10.1097/MNH.0000000000000231.
Han, X., Li, L., Yang, J., King, G., Xiao, Z., & Quarles, L. D. (2016). Counter‐regulatory paracrine actions of FGF‐23 and 1, 25 (OH) 2D in macrophages. FEBS letters, 590(1), 53-67.
Hu, M. C., Kuro-o, M., & Moe, O. W. (2013, March). Renal and extrarenal actions of Klotho. In Seminars in nephrology (Vol. 33, No. 2, pp. 118-129). WB Saunders. doi: 10.1016/j.semnephrol.2012.12.013.
Hui, H., Zhai, Y., Ao, L., Cleveland Jr, J. C., Liu, H., Fullerton, D. A., & Meng, X. (2017). Klotho suppresses the inflammatory responses and ameliorates cardiac dysfunction in aging endotoxemic mice. Oncotarget, 8(9), 15663. doi: 10.18632/oncotarget.14933.
Hum, J. M., O’Bryan, L. M., Tatiparthi, A. K., Clinkenbeard, E. L., Ni, P., Cramer, M. S., ... & White, K. E. (2019). Sustained Klotho delivery reduces serum phosphate in a model of diabetic nephropathy. Journal of Applied Physiology, 126(4), 854-862. doi.org/10.1152/japplphysiol.00838.2018
Hum, J. M., O'Bryan, L., Smith, R. C., & White, K. E. (2017). Novel functions of circulating Klotho. Bone, 100, 36-40. doi:10.1016/j.bone.2016.11.025.
Izbeki, F., Asuzu, D. T., Lorincz, A., Bardsley, M. R., Popko, L. N., Choi, K. M., ... & Farrugia, G. (2010). Loss of Kitlow progenitors, reduced stem cell factor and high oxidative stress underlie gastric dysfunction in progeric mice. The Journal of physiology, 588(16), 3101-3117.
Jung, D., Xu, Y., & Sun, Z. (2017). Induction of anti-aging gene klotho with a small chemical compound that demethylates CpG islands. Oncotarget, 8(29), 46745.
Jüppner H. (2011). Phosphate and FGF-23. Kidney Int. Suppl. 121, 24–27.
Kakareko, K., Rydzewska-Rosolowska, A., Brzosko, S., Gozdzikiewicz-Lapinska, J., Koc-Zorawska, E., Samocik, P., ... & Hryszko, T. (2017). The effect of nephrectomy on Klotho, FGF-23 and bone metabolism. International urology and nephrology, 49(4), 681-688.
Kim, H. K., & Jeong, B. H. (2016). Lack of functional KL-VS polymorphism of the KLOTHO gene in the Korean population. Genetics and molecular biology, 39(3), 370-373.
Kim, J. H., Hwang, K. H., Park, K. S., Kong, I. D., & Cha, S. K. (2015). Biological role of anti-aging protein Klotho. Journal of lifestyle medicine, 5(1), 1.
Kim, Y. C., Byun, S., Zhang, Y., Seok, S., Kemper, B., Ma, J., & Kemper, J. K. (2015). Liver ChIP-seq analysis in FGF19-treated mice reveals SHP as a global transcriptional partner of SREBP-2. Genome biology, 16(1), 268.
Krick S, Baumlin N, Aller SP, Aguiar C, Grabner A, Sailland J, Mendes E, Schmid A, Qi L, David NV, & Geraghty P. (2017). Klotho Inhibits Interleukin-8 Secretion from Cystic Fibrosis Airway Epithelia. Sci. Rep. 7, 14388.
Krick, S., Grabner, A., Baumlin, N., Yanucil, C., Helton, S., Grosche, A., ... & Wells, J. M. (2018). Fibroblast growth factor 23 and Klotho contribute to airway inflammation. European Respiratory Journal, 52(1), 1800236. doi:10.1183/13993003.00236-2018.
Kuro-o, M. (2013). Klotho, phosphate and FGF-23 in ageing and disturbed mineral metabolism. Nature Reviews Nephrology, 9(11), 650-660. doi: 10.1038/nrneph.2013.111
Kurosu, H., Yamamoto, M., Clark, J. D., Pastor, J. V., Nandi, A., Gurnani, P., ... & Shimomura, I. (2005). Suppression of aging in mice by the hormone Klotho. Science, 309(5742), 1829-1833.
Kuznik, B. I., VKh, K., Linkova, N. S., Ryzhak, G. A., Sall, T. S., & Trofimova, S. V. Growth factors of fibroblasts FGF19, FGF21, FGF23 as endocrine regulators of physiologica l functions and geroprotectors. Epigenetic mechanisms of regulation. Uspehi sovremennoj biologii. 2017; 137 (1): 84-99.(i n Russian).
Lim, S. W., Jin, L., Luo, K., Jin, J., Shin, Y. J., Hong, S. Y., & Yang, C. W. (2017). Klotho enhances FoxO3-mediated manganese superoxide dismutase expression by negatively regulating PI3K/AKT pathway during tacrolimus-induced oxidative stress. Cell death & disease, 8(8), e2972-e2972. doi:10.1038/cddis.2017.365.
Lin, W., Zhang, Q., Liu, L., Yin, S., Liu, Z., & Cao, W. (2017). Klotho restoration via acetylation of peroxisome proliferation–activated receptor γ reduces the progression of chronic kidney disease. Kidney international, 92(3), 669-679.
Lin, Y., & Sun, Z. (2015). In vivo pancreatic β-cell–specific expression of antiaging gene klotho: a novel approach for preserving β-cells in type 2 diabetes. Diabetes, 64(4), 1444-1458.
Liu, F., Wu, S., Ren, H., & Gu, J. (2011). Klotho suppresses RIG-I-mediated senescence-associated inflammation. Nature cell biology, 13(3), 254-262.
Liu, L., Liu, Y., Zhang, Y., Bi, X., Nie, L., Liu, C., ... & Yang, K. (2018). High phosphate-induced downregulation of PPARγ contributes to CKD-associated vascular calcification. Journal of molecular and cellular cardiology, 114, 264-275.
Lorenzi, O., Veyrat-Durebex, C., Wollheim, C. B., Villemin, P., Rohner-Jeanrenaud, F., Zanchi, A., & Vischer, U. M. (2010). Evidence against a direct role of klotho in insulin resistance. Pflügers Archiv-European Journal of Physiology, 459(3), 465-473.
Messaoudi, S., Tebibel, S., Beladjila, A. K., Touhami, F. K., & Kabouche, Z. (2019). Anti-hyperlipidemic, Anti-inflammatory and Antioxidant Activities of Citrullus lanatus. World, 8(1), 100-106.
Moskalev, A. A. (2013). Genetics and Epigenetics of aging and longevity. Ecological genetics, 11(1), 3-11.
Mourad, B., Rachid, B., & Sihem, B. (2018). Antioxidant Activity and Phenolic Content of Artemisia Campestris from Two Regions of Algeria. World Journal of Environmental Biosciences, 7(2), 61-66.
Olejnik, A., Franczak, A., Krzywonos-Zawadzka, A., Kałużna-Oleksy, M., & Bil-Lula, I. (2018). The biological role of klotho protein in the development of cardiovascular diseases. BioMed research international, 2018.
Pavlov, S. V., & Belenichev, I. F. (2014). Molecular and biochemical aspects of the neuroprotective effect of the selective estrogen receptor modulator tamoxifen in a model of acute cerebral ischemia. Neurochemical Journal, 8(1), 28-32.
Razzaque, M. S. (2012). The role of Klotho in energy metabolism. Nature Reviews Endocrinology, 8(10), 579.
Richter, B., & Faul, C. (2018). FGF23 actions on target tissues—with and without klotho. Frontiers in endocrinology, 9, 189. doi:10.3389/fendo.2018.00189.
Rosen, E. D., & Spiegelman, B. M. (2000). Molecular regulation of adipogenesis. Annual review of cell and developmental biology, 16(1), 145-171.
Rosidi, A. (2020). The difference of Curcumin and Antioxidant activity in Curcuma Xanthorrhiza at different regions. Journal of Advanced Pharmacy Education & Research| Jan-Mar, 10(1), 15.
Schiff, M., Bénit, P., Jacobs, H. T., Vockley, J., & Rustin, P. (2012). Therapies in inborn errors of oxidative metabolism. Trends in Endocrinology & Metabolism, 23(9), 488-495.
Schumann, G., Liu, C., O’Reilly, P., Gao, H., Song, P., Xu, B., ... & Lepe, M. S. (2016). KLB is associated with alcohol drinking, and its gene product β-Klotho is necessary for FGF21 regulation of alcohol preference. Proceedings of the National Academy of Sciences, 113(50), 14372-14377.
Shen, D., Li, H., Zhou, R., Liu, M. J., Yu, H., & Wu, D. F. (2018). Pioglitazone attenuates aging-related disorders in aged apolipoprotein E deficient mice. Experimental gerontology, 102, 101-108.
Singh, S., Grabner, A., Yanucil, C., Schramm, K., Czaya, B., Krick, S., ... & Brand, M. (2016). Fibroblast growth factor 23 directly targets hepatocytes to promote inflammation in chronic kidney disease. Kidney international, 90(5), 985-996. doi:10.1016/j.kint.2016.05.019/
So, W. Y., Cheng, Q., Chen, L., Evans-Molina, C., Xu, A., Lam, K. S., & Leung, P. S. (2013). High glucose represses β-klotho expression and impairs fibroblast growth factor 21 action in mouse pancreatic islets: involvement of peroxisome proliferator–activated receptor γ signaling. Diabetes, 62(11), 3751-3759.
Sosa-Pineda, B., Chowdhury, K., Torres, M., Oliver, G., & Gruss, P. (1997). The Pax4 gene is essential for differentiation of insulin-producing β cells in the mammalian pancreas. Nature, 386(6623), 399-402.
Sugiura, H., Yoshida, T., Mitobe, M., Yoshida, S., Shiohira, S., Nitta, K., & Tsuchiya, K. (2010). Klotho reduces apoptosis in experimental ischaemic acute kidney injury via HSP-70. Nephrology Dialysis Transplantation, 25(1), 60-68. doi: 10.1093/ndt/gfp451.
Sugiura, H., Yoshida, T., Tsuchiya, K., Mitobe, M., Nishimura, S., Shirota, S., ... & Nihei, H. (2005). Klotho reduces apoptosis in experimental ischaemic acute renal failure. Nephrology Dialysis Transplantation, 20(12), 2636-2645.
Ushigusa, T., Ichinose, K., Sato, S., Michitsuji, T., Shimizu, T., Umeda, M., ... & Kawashiri, S. Y. (2016). Soluble α-klotho is a potential biomarker associated with neuropsychiatric systemic lupus erythematosus. Clinical Immunology, 165, 29-34.
Wang, Y., Kuro‐o, M., & Sun, Z. (2012). Klotho gene delivery suppresses Nox2 expression and attenuates oxidative stress in rat aortic smooth muscle cells via the cAMP‐PKA pathway. Aging cell, 11(3), 410-417.
Wesseling-Perry, K. (2010). FGF-23 in bone biology. Pediatric Nephrology, 25(4), 603-608.
Xu, Y., & Sun, Z. (2015). Molecular basis of Klotho: from gene to function in aging. Endocrine reviews, 36(2), 174-193. doi: 10.1210/er.2013-1079.
Yao, Y., Wang, Y., Zhang, Y., & Liu, C. (2017). Klotho ameliorates oxidized low density lipoprotein (ox-LDL)-induced oxidative stress via regulating LOX-1 and PI3K/Akt/eNOS pathways. Lipids in health and disease, 16(1), 77. doi: 10.1186/s12944-017-0447-0.
Yokoyama, S., Oguro, R., Yamamoto, K., Akasaka, H., Ito, N., Kawai, T., ... & Sugimoto, K. (2019). A klotho gene single nucleotide polymorphism is associated with the onset of stroke and plasma klotho concentration. Aging (Albany NY), 11(1), 104.
Zhang, H., Li, Y., Fan, Y., Wu, J., Zhao, B., Guan, Y., ... & Wang, N. (2008). Klotho is a target gene of PPAR-γ. Kidney international, 74(6), 732-739.
Zhao, Y., Banerjee, S., Dey, N., LeJeune, W. S., Sarkar, P. S., Brobey, R., ... & Choudhary, S. (2011). Klotho depletion contributes to increased inflammation in kidney of the db/db mouse model of diabetes via RelA (serine) 536 phosphorylation. Diabetes, 60(7), 1907-1916.
Zhou, X., & Wang, X. (2015). Klotho: a novel biomarker for cancer. Journal of cancer research and clinical oncology, 141(6), 961-969.
Zhu, Z., Xia, W., Cui, Y., Zeng, F., Li, Y., Yang, Z., & Hequn, C. (2019). Klotho gene polymorphisms are associated with healthy aging and longevity: Evidence from a meta-analysis. Mechanisms of ageing and development, 178, 33-40.